Equilibrium And Advanced Thermodynamics: Balance In Chemical Reactions
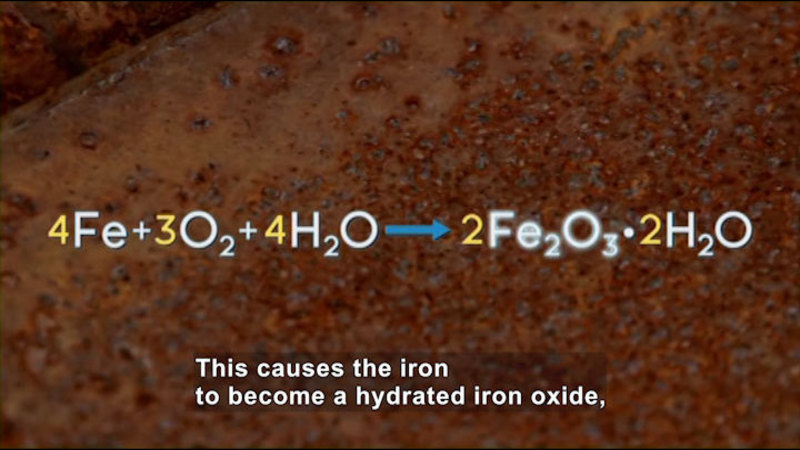
Some chemical reactions happen spontaneously, like metal rusting. Other reactions are non-spontaneous and need to absorb energy in order to occur. Using the Second Law of Thermodynamics, the principle of entropy, and the calculation of Gibbs free energy, scientists can predict which reactions will occur and vary the conditions to make more of the desired products. In equilibrium reactions, both products and reactants are always present. Equilibrium reactions in the human body are essential for life and can be exploited in chemical manufacturing as well. Part of the series Chemistry: Challenges And Solutions.
(Source: DCMP)
Metadata
- Subject:
- Physical Sciences - Science
- Keywords:
- chemistry, science methods
Files 1
-
Equilibrium And Advanced Thermodynamics: Balance In Chemical Reactions

- Type:
- Video
- Format:
- Streaming
- Accommodations:
- English Audio Descriptions - Visual, English Captions - Auditory
- Languages:
- English
- License:
- DCMP Membership
- Author:
- Annenberg
- Length:
- 29 minutes
Collections 1
This resource is part of one or more collections.
-
Chemistry -
 Video
Video
-
 Image
Image
-
 2.5D Tactile Graphic
2.5D Tactile Graphic
-
 PDF
PDF
-
 Text Document
Text Document
-
 Simulation
Simulation

A collection of Chemistry related resources
A collection containing 67 resources, curated by Benetech
-
